Magnesium burns with an intense white flame creating magnesium oxide. Beryllium burns with a sparkly and silvery visual, this forms beryllium There is no trend in which group 2 metals react with oxygen, but as the metals are burnt, they Reaction of beryllium, 2 Be(s)+O 2 (g) 2 BeO(s). When they react with oxygen a solidĬompound is created, called metal oxide, these have a high melting point. Group 2 metals are earth metals which are alkaline. When the reduction agent is oxidised, it loses electrons and the oxidation numberĮxplain the reactions of named group 2 metals with oxygen. Oxidation number will decease when it loses electrons, this occurs when an oxidising agent is Loses electrons and where is has lost oxygen it has been reduced, gaining electrons. Where a compound has gained oxygen, it has been oxidised, it Oxidation numbers can be positive and negative, they’re assigned to an atom to determine its Using oxidation numbers to decide if elements/compounds are oxidised or reduced in given 
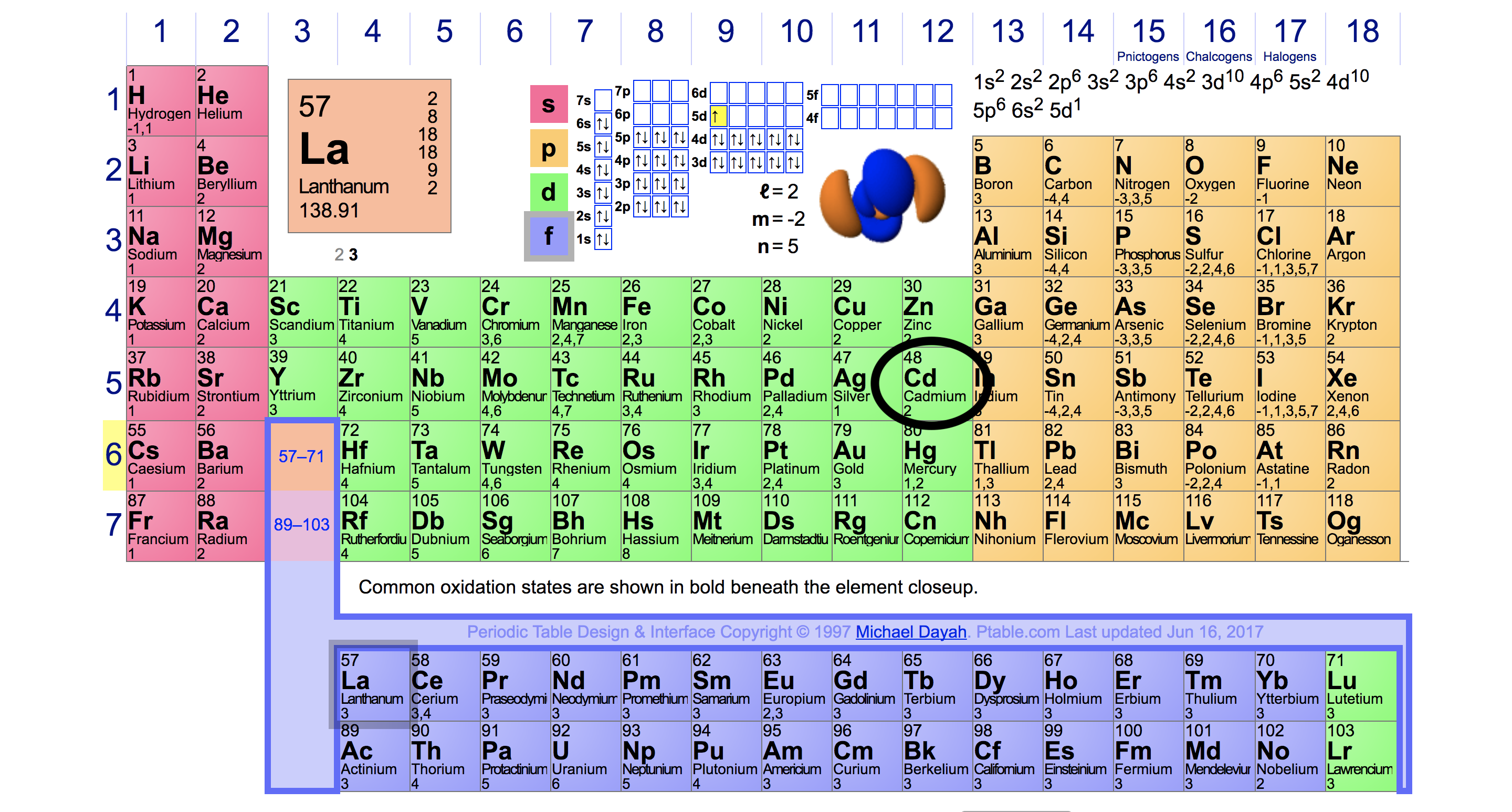
The large jump willĬorrespond to moving from the 3s to the full 2p subshell 1s2 2s2 2p6 3s Last electron in the valence shell therefore sodium belongs to group 1. Showing that the first electron to be removed is the Remove the first electron than the second. Sodium has an atomic number of 11,īetween the first and second ionisation energy there’s a huge jump, which shows it is easier to Successive ionisation energy can be used to predict the electron configuration butĪnalysing the large jumps appear and the number of electrons removed when the large jumpsĪppear then the electron configuration can appear. Electronic configuration can be predicted by its atomic Predicting electron configuration from ionisation energiesĮlectronic configuration is how electrons are arranged in an atom. (Free Printable periodic tables (pdf and PNG) 2023)įigure 3: (First ionisation energy across period 3 2023) These 3 blocks are significant as they explain the chemical properties of the elements as well asįigure 2 Blocks in the periodic table. 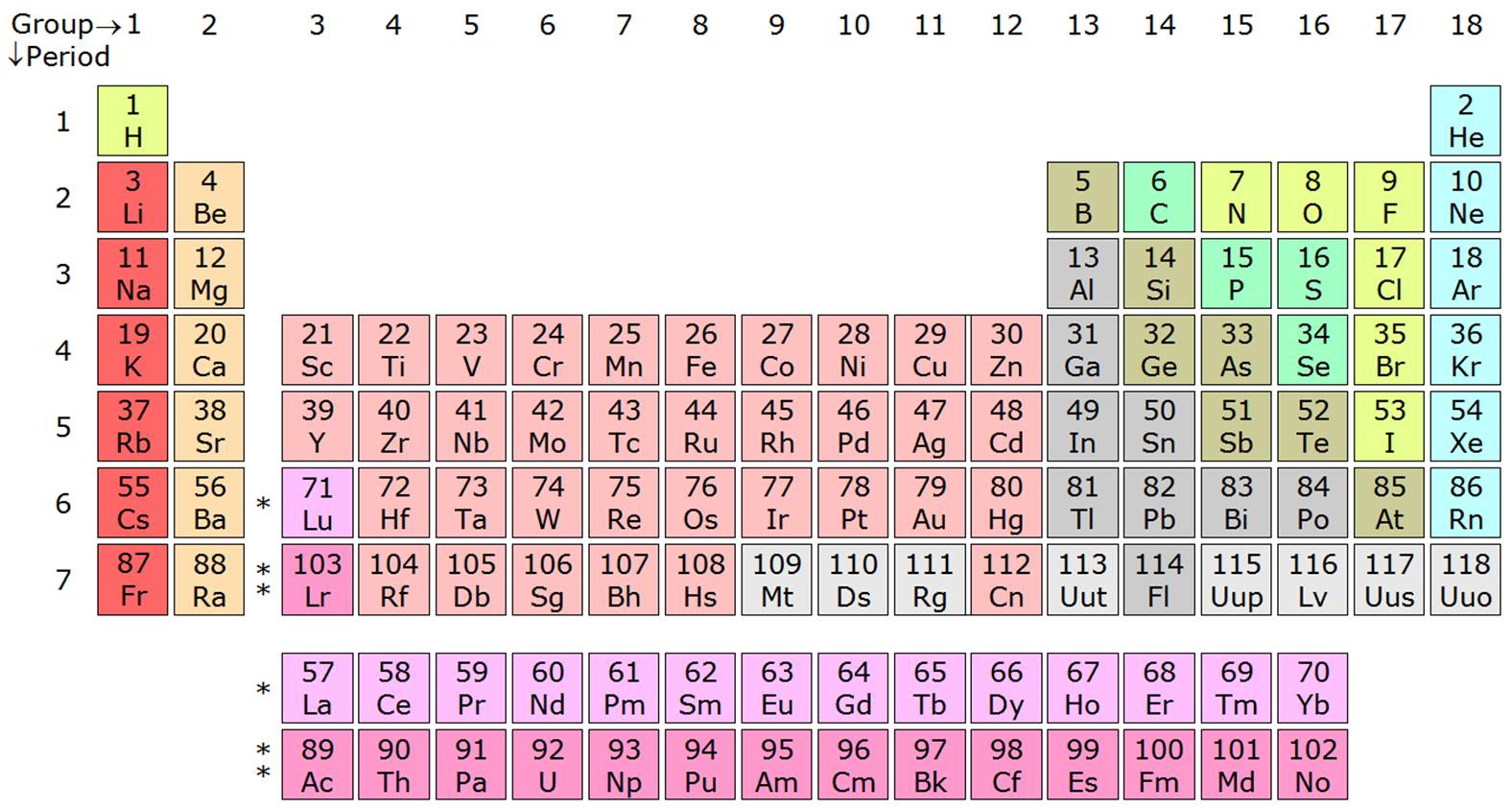
With high melting points and boiling points, with the ability to hold 10 electrons. The D block, consists of transition metals located in the middle of the table. With the ability to gain, loose and share their valence electrons, the P orbitals can hold P block is the last 6 groups of the periodic table non metals, metalloids, and post-transition Melting and boiling point due to weak metallic bonding. These are reactive metals with high ionization enthalpies, having a low The S block elements are highly reactive as the S orbital can only The S block contains group 1 and group 2 and Helium, they’re metals known as alkali metalsĪnd alkaline earth metals. The blocksĮxplain the electronic configuration of the elements as well as their chemical properties. Elements within these blocks areĬategorised in accordance with the orbital occupied by the outermost electron. The periodic table is split into 4 blocks S,P,D and F. Organisation in the table, and allows understanding of chemical reactions in elements.įigure 1: Periodic Table (Free Printable periodic tables (pdf and PNG) 2023)Įxplain the significance of the S, P and D blocks in the periodic table. The element properties, the groups are named, allowing there to be categorising and The overall significance of the main groups and number of the periods is the predictability of Less reactive as it is harder to remove 18 electrons than 1. Highly reactive, it has 1 electron in the outer shell, the noble gases having 18 electrons makes it The group begin with group 1 being alkali metals and ending with noble gases at 18. The ability to readily loose theĮlectrons on the shell contribute to the chemical reactions. The number of shells occupied determineĮach elements atomic radius, its reactivity and ionisation energy. In the outermost shell shows which group it is in. The period number is the number of shells around the nucleus, the number of electrons The periodic table gives an atoms electron configuration based upon its location within the The groups run in vertical columns from 1 to 18.Įlements are put in order of their atomic number, meaning elements with similar propertiesĪppear in the same vertical column or group, properties such as melting point, melting point,ĭensity, and hardness. Horizontal rows and are numbered 1 down to 7. Predicted in the elements thus predicting reactions between each element. The position of the elements within the table enables properties to be predicted. Bethan White | Unit 8 | 15/11/ Contents PageĮxplain the significance of the main groups and numbering of the periods. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
